
Overall, the importance of stoichiometry cannot be overstated. By learning about stoichiometry, students can develop their problem-solving skills and gain a deeper understanding of the underlying principles of chemistry. In addition to its practical applications, stoichiometry is also a fundamental concept in chemistry that helps students to understand the behavior of chemical systems. By predicting the amounts of reactants and products involved in a reaction, we can assess the potential risks and benefits of a given chemical process and make informed decisions about its use. Stoichiometry is also important in the study of environmental chemistry, as it allows us to understand the impact of chemical reactions on the environment. 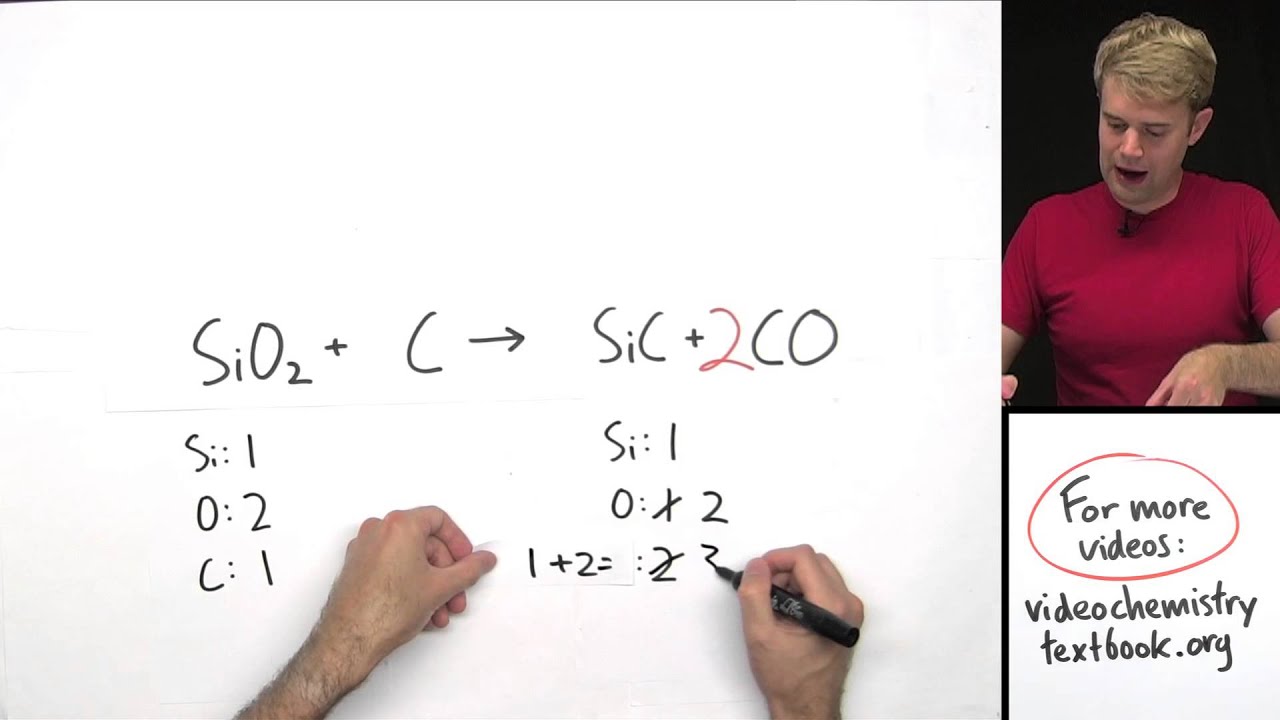
This is critical in the design of industrial processes, where efficiency and cost are key considerations. For example, stoichiometry can be used to determine the amount of reactants needed to produce a certain quantity of a product, or to calculate the yield of a reaction. One of the primary applications of stoichiometry is in the field of chemical engineering, where it is used to design and optimize chemical processes. This branch of chemistry is concerned with the relationships between the quantities of substances involved in chemical reactions, and allows chemists to predict the amounts of reactants and products based on the balanced chemical equation for the reaction. The importance of stoichiometry in chemistry cannot be overstated. Parentheses ( ), square brackets, and braces.Write the symbols of the elements using capital letters for the first letter.Example: To enter the equation 2Na (s) + Cl 2 (g) ⟶ 2NaCl (s), we ignore (s) and (g), writing 2Na+Cl2 = 2NaCl. If the state of the compounds is specified in the equation that you are going to enter, eliminate them from the equation. It is not allowed to specify the state of the compounds, (s) for solid, (g) for gaseous or (l) for liquid.Use the symbol = to separate the reactants from the products.To correctly enter the chemical equation we must follow the following syntax rules: To use the Stoichiometry Salculator you just need to write the chemical equation in the input field and then press the calculate button. The Reaction Stoichiometry Calculator allows you to balance a chemical equation and establish the relationship between the amounts of reactants and products of the reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
